
Electrolysis of water by using a source of renewable energy is an important technology for carbon-free hydrogen production. This reaction takes place in a unit called an electrolyzer. Based on the types of electrolytes and operating conditions, electrolyzers can be classified into three main categories: 1 ) alkaline water electrolyzer (AWE) 2 ) high temperature solid oxide electrolyzer (SOE) 3 ) proton exchange membrane(PEM) electrolyzer.
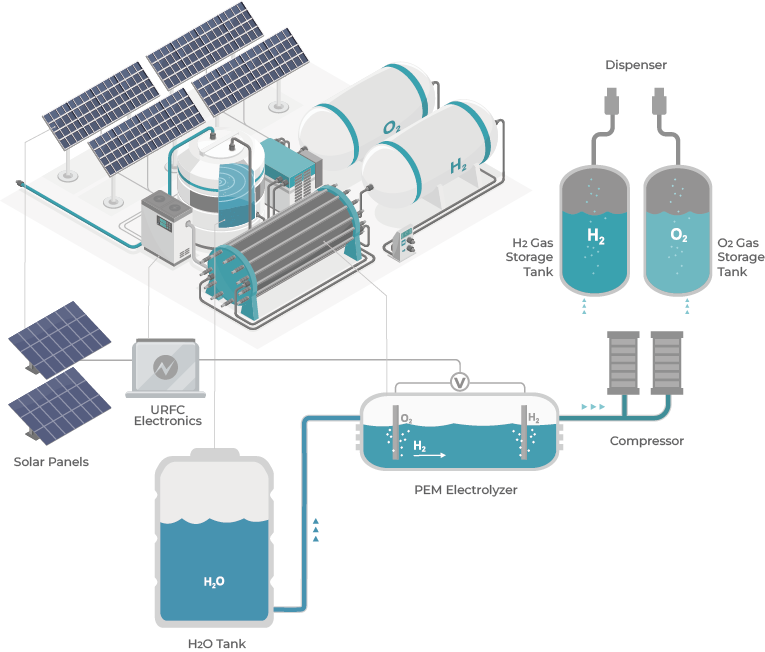
Platinum-based materials are presently recognized as the state-of-the-art electro-catalysts for the cathode side in PEM electrolyzers, which exhibit decent electrochemical activity for the hydrogen evolution reaction (HER) in acidic electrolytes.
In view of cost reduction and high efficiency, our platinum coated titanium mesh electrode is a promising option for long-lasting performance.